Your Guide to Research Quality Control
Think of building a house. You wouldn't just slap on a coat of paint at the end and call it done, right? You’d... by @outrank | Factiii
Your Guide to Research Quality Control
Think of building a house. You wouldn't just slap on a coat of paint at the end and call it done, right? You’d check the foundation, the framing, the plumbing—every single step. That's exactly what **research quality control** is. It’s the constant, careful process of making sure your work is solid from the ground up, so your final conclusions are credible, reliable, and can hold up under pressure.
## Why Research Quality Control Is Your Most Important Experiment

You can think of quality control as applying the scientific method *to your own scientific method*. It’s a deliberate system for catching errors, stamping out bias, and making sure the data you collect is actually trustworthy. Without it, even a groundbreaking idea can rest on a faulty premise, leading to results that are wrong, impossible to replicate, and sometimes, even dangerous.
The fallout from poor quality control can be huge. It can sink projects, waste valuable funding, and tarnish a researcher's reputation. In fields like medicine or engineering, the stakes are even higher—a lack of rigor can directly impact public health and safety.
### The Core Pillars of Research Quality Control
To get it right, quality control really boils down to a few essential components. These pillars are what hold up the integrity of your work, ensuring every stage of your project is handled with precision and care. They form a comprehensive system that protects your research from start to finish.
This table breaks down the core pillars that every researcher should focus on.
### Core Pillars of Research Quality Control
| Pillar | Objective | Example Action |
| :--- | :--- | :--- |
| **Reproducibility** | Ensure others can repeat your experiment and get similar results. | Creating detailed Standard Operating Procedures (SOPs) for every lab protocol. |
| **Accuracy** | Make sure your measurements reflect the true value. | Regularly calibrating all instruments and double-checking data entry. |
| **Traceability** | Maintain a clear, unbroken trail for every data point. | Labeling samples with unique IDs and keeping a meticulous logbook. |
| **Consistency** | Apply methods uniformly across the entire study. | Training all team members on the same protocols to avoid variation. |
Each pillar plays a critical role. When they work together, they build a powerful foundation of trust for your findings.
> "The ability to model human tissues and tumors 'in a dish' enables researchers to better understand how growth responses to culture conditions impact cellular differentiation mechanisms and viability."
This quote really drives home how crucial this is, especially in complex areas like organoid research. With so many variables in 3D cell cultures, strict quality control is the only way to know if your results are real or just random noise. The whole point is to manage those variables so you can have confidence in your conclusions.
### It's More Than Just Avoiding Mistakes
Let’s be clear: solid quality control isn't just about playing defense and preventing errors. It's a proactive strategy for producing work that truly makes an impact. When you're absolutely confident in your data, you can ask bigger questions, collaborate with more assurance, and present your findings with authority.
In an era where data is everywhere and skepticism is high, robust quality control is your best friend. It’s what separates solid, evidence-based science from questionable claims. It’s the unsung hero of every great experiment, ensuring your work doesn't just add to the conversation—it earns the respect it deserves. For any serious researcher, mastering these principles is fundamental to making a meaningful discovery.
## Building Your Foundation for Research Integrity
It's one thing to understand why **research quality control** matters, but it’s another thing entirely to put it into practice. Moving from theory to action means building a practical toolkit—a set of core methods that stop errors before they have a chance to mess up your work. This is where you build the operational muscle to turn quality control from a fuzzy concept into a daily habit for your whole team.
The aim is to create a system so clear and solid that it becomes second nature. Think of a busy restaurant kitchen. Every chef knows their station, every recipe is standardized, and every plate gets checked before it goes out. That systematic approach guarantees every customer gets the same high-quality meal, every time. Your research deserves that same level of precision.
### Develop Clear Standard Operating Procedures
The bedrock of any solid quality control system is the **Standard Operating Procedure (SOP)**. An SOP isn't just a simple checklist. It's a detailed, step-by-step instruction manual that spells out exactly how to perform a specific task, leaving no room for guesswork. By documenting your processes, you ensure everyone does the task the exact same way, no matter who's doing it.
For example, an SOP for preparing a cell culture would break down everything:
* **Materials List:** The specific reagents, media, and labware needed.
* **Step-by-Step Instructions:** Every single action, from warming the media to the precise incubation time and temperature.
* **Safety Precautions:** What personal protective equipment is required and how to handle materials safely.
* **Documentation Requirements:** Exactly how to label the culture flask and record the process in the lab notebook.
Creating these documents forces you to think through every step, often shining a light on weak spots or potential errors you might have missed. A well-written SOP is your best defense for ensuring your results are reproducible and consistent.
This process flow shows the basic sequence of a quality control framework, from setting the initial standards all the way to final approval.
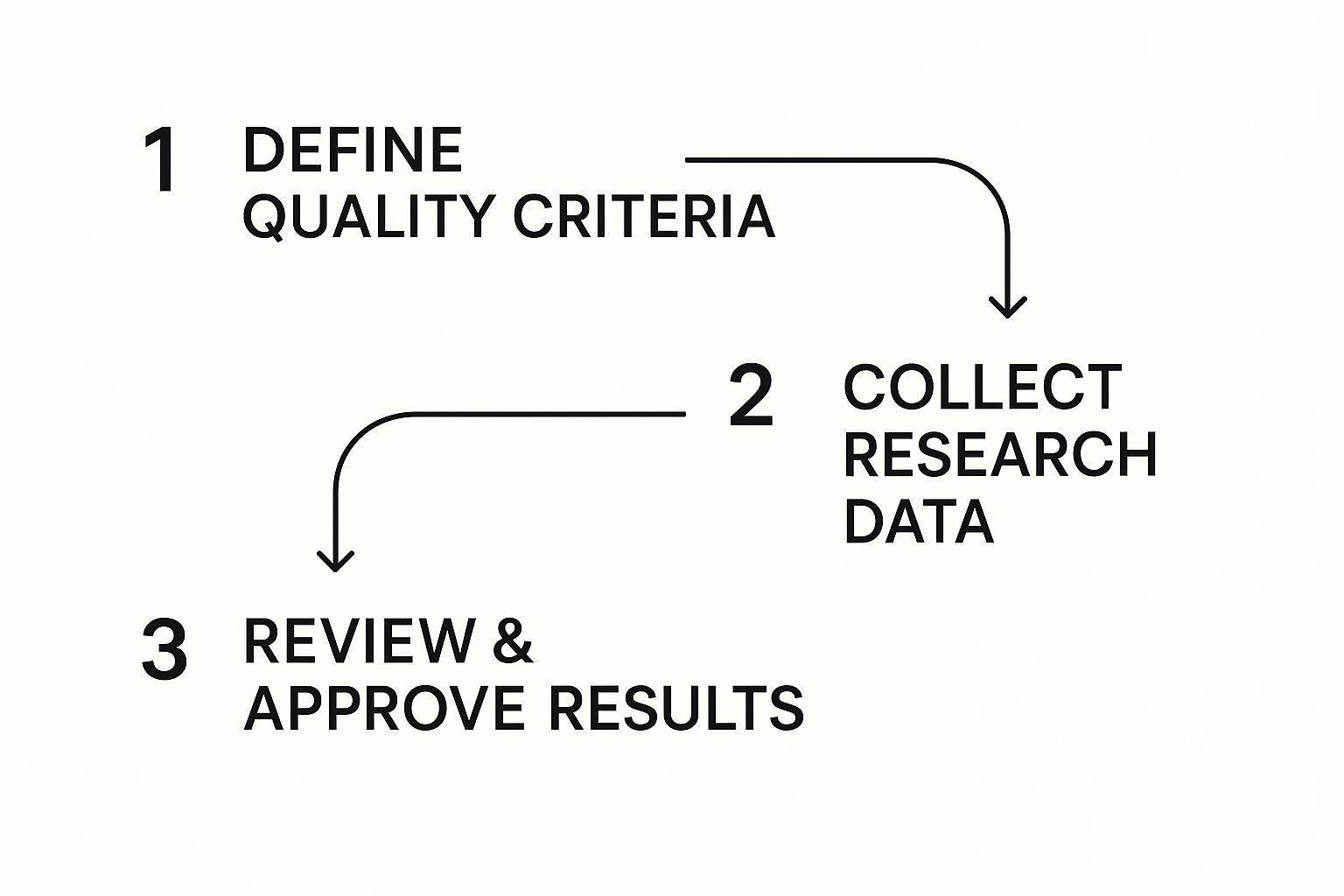
As the visual shows, quality isn't an accident. It's a planned journey with clear checkpoints that protect the integrity of your work from start to finish.
### Master Data Validation Techniques
Even with perfect procedures, mistakes can still happen, especially when people are entering and managing data. This is where data validation comes in. These methods are designed to catch errors at the source, saving you the headache of finding a critical flaw months down the road.
One of the simplest and most effective methods is **double-data entry**. It’s exactly what it sounds like: two different people enter the same data into two separate files. Then, you use software to compare the files and flag any differences. It's a low-tech solution that pays huge dividends in data integrity.
> A study in a clinical trial found that double-data entry caught and corrected errors in **2.6%** of all data fields. It might feel redundant, but that small time investment can be the difference between sound and flawed conclusions.
Another great technique is to build validation rules right into your spreadsheets or databases. For instance, you can:
* **Set Range Checks:** Limit a field to only accept numbers within a logical range (like pH values between 0 and 14).
* **Use Data Type Constraints:** Make sure a column only accepts certain data types, like dates or whole numbers.
* **Create Dropdown Lists:** Use preset lists for categories to avoid typos or variations (e.g., "Male," "Female" instead of "M," "f," or "guy").
These small, automated checks act as your first line of defense, keeping your dataset clean and reliable from the very beginning.
## Running Your Lab Like a Well-Oiled Machine

A top-notch **research quality control** system isn't just a binder of rules collecting dust on a shelf. It's the living, breathing workflow that’s woven into the fabric of your daily operations. I like to think of it as a symphony orchestra. You can have the most talented musicians in the world, but without a conductor keeping everyone in sync, you just have noise.
In any modern research setting, that harmony comes from integrating a few key processes. When done right, they turn quality from a lofty goal into a natural, almost automatic outcome. It's about building a system that anticipates problems before they happen, ensuring every single step of your research is consistent and trustworthy.
### Audit Management: The Conductor's Baton
If your lab is the orchestra, then audit management is the conductor's baton. Audits, whether they're internal or from an outside group, are really just systematic reviews to check if you’re actually doing what you say you’re doing. It’s not about pointing fingers; it's about finding those little opportunities to get better.
For instance, a simple internal audit might show that different team members are using slightly different versions of the same protocol. That's not a failure—it's a critical insight! It tells you that your document management might need to be tightened up, or maybe it’s time for a quick refresher training session.
Regular audits give you a clear, honest snapshot of your operations. They make sure every process is on track and meets the necessary standards.
### Document Control: The Official Sheet Music
For an orchestra to play a piece perfectly, every musician needs to be reading from the same, correct version of the sheet music. That's exactly what **document control** does for a research team. It's the system you use to manage the creation, approval, distribution, and archiving of all your important documents.
This covers everything from your Standard Operating Procedures (SOPs) and lab protocols to equipment calibration logs and training records. A solid document control system ensures a few key things:
* **Clear Version Control:** Only the latest, approved version of any document is available. This simple step prevents someone from accidentally using an outdated procedure.
* **Controlled Access:** Team members can easily find the documents they need, but no one can make unauthorized changes.
* **Tracked Changes:** Every revision to a document is logged, creating a transparent history of who changed what, when, and why.
Without this, you’re inviting chaos. Imagine one researcher following an old protocol while another uses a newer, updated one. Your results would be impossible to compare, and their validity would immediately be in question.
> Effective document control isn't just boring administrative work; it's a cornerstone of reproducibility. It guarantees that the "how" behind your data is always clear, consistent, and defensible.
This kind of organization is getting easier thanks to specialized software. The global Quality Management Software (QMS) market was valued at around **USD 11.14 billion** in 2024 and is on track to nearly double by 2030. This boom shows a clear global shift toward using centralized systems for everything from document management to supplier quality. If you're interested, you can [explore the full market analysis](https://www.fortunebusinessinsights.com/quality-management-software-qms-market-102555) to see where the industry is headed.
### Supplier Quality: The Instrument Makers
At the end of the day, your research is only as good as the tools and materials you use. **Supplier quality oversight** is all about making sure the reagents, equipment, and services you buy actually meet your standards. A bad batch of antibodies or a poorly calibrated instrument can completely invalidate months of hard work.
This means your quality control has to extend beyond the four walls of your lab. It involves:
* **Vetting Suppliers:** Carefully choosing vendors who have a proven track record for quality and reliability.
* **Setting Clear Specs:** Defining *exactly* what you need when you place an order, leaving no room for error.
* **Inspecting Incoming Goods:** Taking a moment to verify that what you received is what you ordered and that it’s up to snuff.
When you bring audit management, document control, and supplier oversight together, you create a complete, holistic workflow. It’s a system where every part—your people, your processes, and your materials—works together in concert to produce research that you can stand behind.
## How Technology Is Revolutionizing Quality Control
For decades, the gold standard for quality control was a human with a clipboard. Manual checks and balances were the only game in town, but they've always had their limitations. People get tired, make mistakes, and frankly, checking massive datasets by hand is slow and tedious.
Now, a new wave of technology is completely changing the game. We're moving beyond simply catching mistakes after they happen. The goal today is to build systems that prevent errors from ever occurring.
Think about modern lab equipment that automatically logs every result into a central database. Just like that, the risk of a simple typo contaminating an entire dataset is gone. It’s a fundamental shift from looking backward at what went wrong to actively ensuring things go right in real time.
### The Rise of Automation and AI
The biggest leaps forward are thanks to **automation** and **artificial intelligence (AI)**. Automation is brilliant at handling repetitive, rule-based jobs with flawless consistency—something humans just can't match. AI takes it a step further by analyzing complex patterns to make smart predictions, essentially giving your research team a tireless digital assistant.
Together, these technologies create a powerful new quality framework. Automation handles the grunt work with perfect precision, while AI provides the high-level analysis. For a deeper dive into this, you can [review an in-depth look at emerging trends in quality control](https://metrology.news/the-future-of-quality-control-a-look-at-upcoming-trends/) to see how Industry 4.0 is connecting devices for continuous monitoring.
This image from Metrology News shows just how interconnected modern quality systems have become.
You can see how a single system pulls in data from all over—production lines, supplier reports, even customer feedback—to get a complete, 360-degree view of quality.
### What This Looks Like in Practice
So, how does this all play out in a real lab? The applications are incredibly diverse and are already making a huge impact.
Here are a few ways technology is sharpening **research quality control**:
* **Automated Data Validation:** Forget spending hours squinting at spreadsheets. Automated scripts can now scan entire datasets in seconds, flagging outliers, missing values, or formatting mistakes. This frees up researchers to do what they do best: analyze the data, not just clean it.
* **Predictive Equipment Maintenance:** Smart sensors can now monitor lab equipment and predict when a part is likely to fail. This proactive approach helps avoid those catastrophic breakdowns that can ruin an experiment and wipe out valuable data.
* **AI-Powered Literature Review:** Instead of spending weeks in the library, AI tools can sift through thousands of research papers in minutes. They can pinpoint relevant studies, flag conflicting findings, and help researchers build a much stronger foundation for their work.
> By automating routine checks and using AI for complex analysis, research teams can achieve a level of precision and efficiency that was previously impossible. The focus shifts from merely catching errors to proactively building quality into every step of the research lifecycle.
### Key Tools Driving the Change
A few specific technologies are leading the charge, each offering unique ways to help research teams manage complex work with more confidence.
1. **Laboratory Information Management Systems (LIMS):** Think of a LIMS as the central nervous system for your lab. It automates workflows, tracks every sample, manages instrument calibration, and keeps a detailed audit trail of every single action.
2. **Electronic Lab Notebooks (ELNs):** ELNs are the modern replacement for paper notebooks. They create a secure, searchable, and collaborative digital record of all your work. They help standardize how data is entered, protect your findings with timestamps, and make sharing protocols a breeze.
3. **Collaborative Platforms like Factiii:** New platforms are also emerging to tackle quality from a different angle. Tools like [Factiii](https://factiii.com) create a community-driven space where researchers can help verify claims and data. This fosters a culture of transparency that goes beyond a single lab, boosting integrity across the entire field.
In the end, technology isn't here to replace researchers—it's here to empower them. By taking over the tedious tasks and spotting patterns humans might miss, these tools give scientists the freedom and confidence to pursue groundbreaking discoveries.
## Quality Control in Action: A High-Stakes Case Study

It’s one thing to talk about theories and workflows, but the true test of any **research quality control** system is how it holds up under pressure. And there's no better example of a high-pressure environment than In Vitro Diagnostics (IVD), where a single lab result can change someone's life forever.
In this field, quality control isn't just a good idea—it's an absolute necessity for keeping people safe. It’s the real-world application of quality principles at their most critical.
When it comes to IVD, every test, from a basic blood sugar check to a sophisticated genetic screening, has to be provably accurate. There's simply no margin for error. A wrong result could lead to a misdiagnosis, a flawed treatment plan, or immense emotional turmoil for a patient. This is where a specific tool, the "control material," becomes the hero of the story.
### The Role of Control Materials
Think of it like this: if you’re baking a cake, you don’t just trust that your oven is at the right temperature. You might use a separate, reliable oven thermometer to double-check it. In an IVD lab, control materials are that trusted thermometer.
A **control** is basically a sample that has a known, pre-set value. Labs run these controls right alongside patient samples to make sure their equipment and chemicals are performing exactly as they should.
If the control gives the expected result, the lab knows the patient results are trustworthy. If it doesn't, the entire batch of tests is thrown out, and they start over until the problem is solved. This simple, repetitive check is the bedrock of quality in diagnostics.
The financial numbers back this up. The global IVD quality control market was valued at an enormous **USD 1.3 billion** in 2023, with control materials making up more than half of that value. You can [discover more about the growing IVD quality control market](https://www.gminsights.com/industry-analysis/ivd-quality-control-market) to see just how essential these materials are.
### Ensuring Complete Traceability
It's not just about getting the right answer; it's also about proving how you got it. IVD labs have to maintain a rock-solid chain of evidence for every sample, a practice known as **traceability**.
From the moment a blood sample is taken to the second a report lands on a doctor's desk, every single step is documented, timestamped, and connected. This meticulous record-keeping ensures a few critical things:
* **Sample Integrity:** No mix-ups. Every sample is uniquely identified and tracked from start to finish.
* **Process Transparency:** There's a clear audit trail showing who did what, which machine was used, and what chemicals were involved.
* **Audit Readiness:** If anyone ever questions a result, the lab can immediately produce a complete history of that sample's journey.
> In high-stakes environments like clinical diagnostics, quality control isn't an abstract goal; it's a series of concrete, repeatable actions. The system is designed with the explicit understanding that a human life could depend on its rigor.
This case study is a perfect illustration of how a strong quality framework turns abstract ideas into life-saving actions. By using control materials to confirm accuracy and maintaining detailed records for traceability, the IVD industry builds a system that doctors and patients can trust. It’s a powerful reminder that when research impacts people’s lives, getting quality control right is the most important job there is.
## Your Top Research Quality Control Questions, Answered
Even when you know the principles, putting **research quality control** into practice can feel like a huge task. It's totally normal for questions and roadblocks to pop up when you move from theory to your actual day-to-day work.
This section is your go-to guide for those common "what if" and "how do I" moments. Think of it as a troubleshooter for the practical side of research, designed to help you handle the real-world issues that can trip up even the most careful teams.
### What’s the Difference Between Quality Assurance and Quality Control?
It’s really common to hear these two terms used as if they mean the same thing, but they’re actually two sides of the same coin. Getting the distinction is crucial for building a truly solid quality system.
Let’s use a simple analogy: imagine you’re running a bakery famous for its amazing cakes.
* **Quality Assurance (QA)** is everything you do *before* you even start baking. It's perfecting the recipe, finding the best flour supplier, training your staff on exactly how to mix the batter, and making sure the ovens are always at the right temperature. QA is all about being proactive—it’s designing a great system to *prevent* problems from happening in the first place.
* **Quality Control (QC)** is what happens *after* the cake is baked. It's that final taste test to make sure every single cake is just right—the flavor, the texture, everything. QC is reactive; it’s about *finding* any defects in the final product before it goes out the door.
You can’t have one without the other. A perfect recipe (QA) is useless if the baker messes up, but tasting every cake (QC) is a huge waste of time if your core process is broken. In research, QA is about designing strong protocols, while QC is the hands-on check that confirms those protocols were actually followed.
### How Can a Small Lab Implement Effective QC on a Budget?
This is a big one. So many researchers in smaller labs or startups assume that proper quality control is a luxury, something that requires a massive budget and fancy software. But that's just not true.
Honestly, the most powerful QC tools aren’t expensive systems; they're discipline and good habits. You can get incredible results by focusing on simple, low-cost practices that have a huge impact. The trick is to weave documentation and systematic checks right into your team's daily rhythm.
Here are a few things you can do right now that cost nothing:
* **Standardize File Naming:** Seriously. Just agree on a simple, logical way to name files and folders. This tiny step prevents so much confusion later on.
* **Use a "Buddy System":** Make it a habit for a colleague to do a quick double-check on critical steps, like data entry or key calculations. It’s a simple layer of verification that catches countless small errors.
* **Try Free Version Control:** Tools like [Git](https://git-scm.com/) are free and fantastic for tracking changes in code, analysis scripts, and even manuscripts. You get a clear, transparent history of everything you do.
* **Keep Detailed Digital Notebooks:** Whether you use a fancy Electronic Lab Notebook (ELN) or just a well-organized Word doc, having a clear, searchable record of your experiments is priceless.
> The heart of good quality control isn’t about how much you spend. It’s about building a culture of meticulousness. Consistency is always more valuable than expensive software.
### What Are the First Steps to Improving My Team's Quality Control?
Trying to fix your entire quality system all at once is a surefire way to get overwhelmed and give up. The real secret to making improvements that stick is to start small. Get one win under your belt, and then build from there.
Your very first move should be getting the team involved. Call a quick meeting and ask a simple question: **"What's the one thing in our workflow that causes the most errors or frustration?"**
Then, just listen. More often than not, the answer will be something small and obvious that everyone already knows is a problem—like messy sample labels or a chaotic shared drive.
Once you’ve nailed down that one pain point, focus all your energy on fixing it. For instance, if data storage is the issue, work together to design a simple folder structure and pick one central, backed-up spot for all project data. Done.
Another fantastic starting point is to write a Standard Operating Procedure (SOP) for your most common or most critical experiment. By writing it as a team, you make sure it's practical and you get buy-in from the people who will actually use it every day. Nailing one or two small, visible improvements like this creates the momentum you need to take on the bigger challenges down the road.
### What Software Tools Can Help with Research Quality Control?
While good habits are your foundation, the right tools can definitely make your life easier, especially as your research gets more complex. Lots of great software is out there designed specifically to tackle the headaches of **research quality control**.
Here’s a quick look at the types of tools that can help:
| Tool Category | What It Does | How It Helps with QC |
| :--- | :--- | :--- |
| **Electronic Lab Notebooks (ELNs)** | Keeps digital records | Creates standard data entries, provides clear audit trails, and protects your work with timestamps. |
| **LIMS** | Manages samples & workflows | Automates tracking from start to finish, which cuts down on mix-ups and ensures every sample is accounted for. |
| **Statistical Software (R, SPSS)** | Analyzes data | Has built-in tools for validating data, spotting outliers, and creating analysis scripts you can run again and again. |
| **Quality Management Software (QMS)**| Manages the whole system | Puts document control, SOPs, audits, and corrective actions all in one place for a unified approach. |
Even a simple spreadsheet with good data validation rules can be a game-changer. The key is to pick a tool that matches your team's needs and scale. The right software should reduce the administrative busywork, freeing you up to focus on what really matters: the research itself.
---
At **Factiii**, we believe verifiable information is the bedrock of all meaningful research. Our community-driven platform empowers researchers, journalists, and students to build a more transparent and accountable information ecosystem. By allowing users to submit claims with supporting data and source references, we foster a collaborative environment where facts are scrutinized and quality is a shared responsibility. Strengthen your research by joining a community dedicated to truth. [Discover more at factiii.com](https://factiii.com).